WASHINGTON, Jan. 26, 2023 /PRNewswire/ — Today, the Pharmaceutical Research and Manufacturers of America (PhRMA) released a new report covering 163 medicines in development that are targeting common mental illnesses, all of which are in clinical trials or awaiting review by the U.S. Food and Drug Administration (FDA).
Mental illness represents a broad spectrum of health conditions affecting mood, thinking and behavior. These conditions include depression, bipolar disorder, schizophrenia, substance use disorder, anxiety disorders, eating disorders, obsessive compulsive disorder and post-traumatic stress disorder, among others. Each of these illnesses often varies in its degree of severity, ranging from mild to moderate to severe, and may occur together. In severe cases, mental illnesses can cause serious functional impairment and substantially interfere with or place limits on life activities –– these are considered serious mental illnesses (SMI). Regardless of severity, mental illness can often get in the way of one’s daily activities, personal and professional relationships and limit one’s quality of life.
Fortunately, hope is on the horizon as biopharmaceutical research companies have made tremendous strides towards understanding mental illnesses and developing innovative medicines. By leveraging a growing scientific understanding of the brain, biopharmaceutical researchers are seeking to develop new treatments and bring therapeutic advances for patients who are not helped by current standards of care or for those who may experience negative side effects.
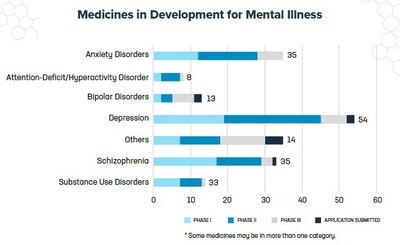
The promise of the pipeline is represented across a wide range of mental illnesses, including:
- 54 for depression, which affects 8.4% of adults and 17% of adolescents aged 12 to 17 in the U.S.
- 35 for schizophrenia, which affects less than 1% of U.S. adults.
- 35 for anxiety disorders. More than a third of adults in the U.S. report experiencing anxiety during their lifetime.
- 33 for substance use disorders. Over 40 million Americans ages 12 and older—or 14.5% of the U.S. population—reported having a substance use disorder in 2020.
- 13 for bipolar disorder which affects about 2.8% of adults and 2.9% of adolescents in the U.S.
- 8 for attention-deficit/hyperactivity disorder (ADHD) which is one the most common childhood disorders with a lifetime prevalence of 11.0% in children ages four to 17 and 8.7% in adolescents ages 13 to 18. ADHD also affects 4.4% of adults in the U.S.
While mental illnesses can impact anyone at any time, not everyone has adequate access to needed care. In fact, more than half of U.S. adults with a mental illness do not receive any treatment at all. Health care access insecurity for patients with mental illness is often driven by coverage and access restrictions. PhRMA’s recent Patient Experience Survey found that patients are increasingly experiencing barriers to adequate care despite having insurance. The survey found:
- 61% of insured Americans managing a mental health condition and taking prescription medicines are concerned the medicines they are prescribed will be subject to insurance formulary exclusion, compared to just 47% of insured Americans taking prescription medicines.
- 56% of insured Americans managing a mental health condition and taking prescription medicines are concerned they would have to wait for their insurer to provide prior authorization for a medicine their doctor prescribed, compared with 42% of insured Americans taking prescription medicines.
- 33% of insured Americans managing a mental health condition have difficulty understanding, anticipating and navigating their insurance coverage, compared with 22% of insured Americans overall.
- At the same time, 94% of insured Americans managing a mental health condition want more predictability in their out-of-pocket costs so that they know how much they will pay for things like prescription drugs every month.
The alarming pace by which rates of mental illness and addiction are increasing across the country demands a comprehensive and multifaceted strategy to stem the tide of these crises. Unfortunately, our health care system is not equipped to handle these increasing trends and ensure patients can access the care they need. In order to fully address the growing mental health challenge in the United States, additional policy reforms are needed that focus on the significant barriers to accessing quality care for patients with these devastating diseases. The biopharmaceutical industry supports policies to:
- Reduce the stigma of mental illness and substance use disorders and bolster our behavioral health workforce.
- Improve access to the comprehensive range of treatment services needed to successfully manage these conditions and make insurance work as intended.
- Greater enforcement of mental health parity laws.
- Leverage and expand the appropriate use of telehealth, while ensuring safeguards to protect the quality of patient care.
- Improve care coordination and delivery through new quality care models.
America’s biopharmaceutical research companies are committed to addressing the stigma of mental illness and researching and developing medicines to improve the health and wellbeing of those struggling with these illnesses. The medicines in development today represent the continued commitment of our industry to advance new treatment options for a wide range of mental illnesses and the diverse patient populations impacted by them.
Read the full report here.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/new-phrma-report-more-than-160-medicines-in-development-for-mental-illness-301731277.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/new-phrma-report-more-than-160-medicines-in-development-for-mental-illness-301731277.html
SOURCE Pharmaceutical Research and Manufacturers of America (PhRMA)