TOKYO, March 30, 2022 /PRNewswire/ — The Global Health Innovative Technology (GHIT) Fund announced today a total of approximately 740 million yen (US$6.4 million*) to invest in three partnerships to develop new lifesaving tools for malaria.** (Appendix 1 & 2)
“We are very pleased to invest in new product development for malaria. Existing innovations alone are not sufficient to combat malaria,” said GHIT CEO Osamu Kunii. “Continuous R&D efforts for new innovations are essential for malaria elimination. We hope that these new innovations will be important tools in the malaria elimination strategy and bring greater impact for patients.”
Preclinical development of a monoclonal antibody to prevent P. falciparum malaria
GHIT will invest 539 million yen (US$4.7 million) for the development of monoclonal antibodies (mAbs) for malaria prevention.. The project is enabled by access to a panel of potent anti-CSP mAbs, identified from volunteers who were protected against P. falciparum challenge following immunization with RTS,S/AS01 (RTS,S), the world’s first malaria vaccine recommended for use in young children living in areas of moderate to high malaria transmission by the World Health Organization (WHO). RTS,S functions primarily by inducing high titer antibodies to the invariant repeat region of the P. falciparum CS protein. Highly potent monoclonal antibodies (mAbs) in protective antisera are likely to be promising tools for malaria intervention.
Development of a Plasmodium vivax multistage vaccine effective both for protection and transmission locking
GHIT will invest 69.8 million yen (US$604K) in the development of a highly effective and durable next-generation multistage/multispecies malaria vaccine that is effective against both pre-erythrocytic stage and sexual-stage P. falciparum and P. vivax parasites.
Hit-to-Lead development of novel Astellas compounds with antimalarial activity
Finally, GHIT will invest 131 million yen (US$1.1 million) in identification of lead compound series with antimalarial activity that has clear potential for further development and progression to lead optimization. Previous project supported by GHIT, four novel hit compounds with antimalarial activity were identified from screening of Astellas’s 20,000 compounds. The compounds will be designed to explore both structure activity relationships and scope for structural modification to improve the compound profile such as potency, DMPK, and safety.
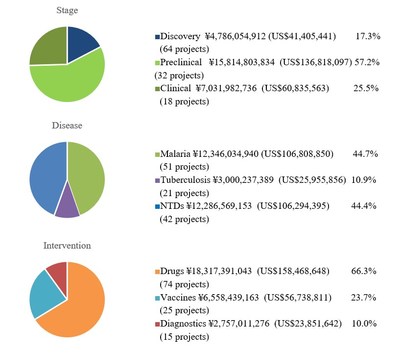
As of March 31, there are 61 ongoing projects, including 28 discovery, 21 preclinical and 12 clinical trials, in the GHIT portfolio (Appendix 3). The total amount of investments since 2013 is 27.6 billion yen (US$239 million).
* USD1 = JPY¥115.59, the approximate exchange rate on February 28, 2022.
** These awarded projects were selected from a number of proposals to the RFP2021-002 for Target Research Platform, Screening Platform, Hit-to-Lead Platform, and Product Development Platform, which was open for applications from June 2021 to January 2022. The GHIT board conducted in February 2022 approved these new investments.
The GHIT Fund is a Japan-based international public-private partnership fund (PPP) between the Government of Japan, multiple pharmaceutical companies, the Bill & Melinda Gates Foundation, the Wellcome, and the United Nations Development Programme (UNDP). The GHIT Fund invests and manages an R&D portfolio of development partnerships aimed at neglected diseases, such as malaria, tuberculosis and neglected tropical diseases that afflict the world’s vulnerable and underserved populations. The GHIT Fund mobilizes the Japanese industry, academia, and research institutes to create new drugs, vaccines, and diagnostics for malaria, tuberculosis, and neglected tropical diseases, in collaboration with global partners.
Appendix.1 New Investments
| ID/Status | Project Title | Collaboration Partners | Disease/Intervention | Stage | Awarded Amount |
| G2021-111 New project | Preclinical development of a monoclonal antibody to prevent P. falciparum malaria | Ehime University, Eisai Co., Ltd., GlaxoSmithKline, PATH | Malaria Drug | Lead Optimization | ¥538,661,663 (US$4,660,106) |
| H2021-201 New project | Hit-to-Lead development of novel Astellas compounds with antimalarial activity | Astellas Pharma Inc., TCG Lifesciences Private Limited. (TCGLS), Medicines for Malaria Venture (MMV) | Malaria Drug | Lead Identification | ¥131,743,584 (US$1,139,749) |
| T2021-256 New project | Development of a Plasmodium vivax multistage vaccine effective both for protection and transmission blocking | Hokkaido University, Jichi Medical University, Kanazawa University, Kyoto University, University of Toyama, University of Cambridge, Instituto Leônidas & Maria Deane (ILMD) and The Fundação de Medicina Tropical Doutor Heitor Vieira Dourado (FMT-HVD) | Malaria | Antigen Identification | ¥69,817,000 (US$604,006) |
*All amounts are listed at the exchange rate of USD1 = JPY¥115.59, the approximate exchange rate on February 28, 2022.
Appendix.2 Project Details
G2021-111
| Project Title | Preclinical development of a monoclonal antibody to prevent P. falciparum malaria |
| Collaboration Partners | Ehime University, Eisai Co., Ltd., GlaxoSmithKline, PATH |
| Disease | Malaria |
| Intervention | Drug |
| Stage | Lead Optimization |
| Awarded Amount | ¥538,661,663 (US$4,660,106) |
| Status | New project |
| Summary | [Project objective] The objective of this project is to complete preclinical development work to support future IND submission for a proof-of-concept clinical trial including controlled human malaria infection (CHMI). The long-term goal of this project is to secure a WHO policy recommendation and financing for a monoclonal antibody that prevents P. falciparum malaria in young children, and potentially pregnant women, living in areas of seasonal transmission in sub-Saharan Africa. 1) Manufacture a cell bank for the lead mAb production, and demonstrate the efficacy of the lead mAb in a mouse challenge model 2) Complete process development and manufacture the lead mAb suitable for a GLP toxicology study 3) Complete a pre-IND meeting with the US FDA 4) Complete preliminary Cost-of-Goods (COGs) analysis for anti-malaria infection mAbs, and develop an initial integrated product development and policy plan (iPDPP).
[Project design] The project is built on successful completion of prior work including 1) in vitro and in vivo screening for a lead panel of mAbs; 2) engineering the lead panel mAbs to enhance potency and serum stability, and to minimize development risk identified by in silico analyses; 3) minimizing risk of selected mAbs with cross-reactivity to human tissues by early screening for binding to human tissues and human membrane proteins; 4) confirming in vivo protective efficacy and pharmacokinetics of the selected mAbs in mouse models; 5), initiate cell line development and select one to advance; and 6) established capabilities of mAb production process development, optimization, and production. The proposed work will start with production and release of a pre-master cell bank for production of the mAb of final selection. The manufacture process for the mAb will be developed and optimized to confirm conditions for production of the mAb lot suitable for GLP toxicity studies, and to enable future scale up of production for clinical trial materials. In addition, formulation development will be conducted to enable stability of the mAb at high concentration to accommodate potential subcutaneous injection. The mAb materials produced during process development and the mAb lot for the GLP toxicity studies will be evaluated in mouse challenge model to confirm protective efficacy. A pre-IND meeting will be conducted with the US FDA. We will also conduct IND-enabling GLP-toxicology studies to evaluate repeated dose toxicity and tissue-cross reactivity of the mAb. In parallel, a COGs analysis will be conducted with information on methods of production, product configuration (vial type and size), and a competitive landscape analysis will be conducted with information on mAbs in development for malaria prevention. Finally, an iPDPP will be drafted for alignment among clinical, regulatory, manufacturing, and policy strategies for global health deployment of mAbs and alike. |
| Project Detail | https://www.ghitfund.org/investment/portfoliodetail/detail/201/en |
H2021-201
| Project Title | Hit-to-Lead development of novel Astellas compounds with antimalarial activity |
| Collaboration Partners | Astellas Pharma Inc., TCG Lifesciences Private Limited. (TCGLS), Medicines for Malaria Venture (MMV) |
| Disease | Malaria |
| Intervention | Drug |
| Stage | Lead Identification |
| Awarded Amount | ¥131,743,584 (US$1,139,749) |
| Status | New project |
| Summary | [Project objective] The objective of the project is to identify at least 1 novel compound series meeting MMV Early Lead Criteria (1) that has clear potential for further development and progression to Lead Optimization.
[Project design] The project will consist of two phases. The first 6-month phase will involve the synthesis and profiling of small set of compounds designed around each of the four hit compounds identified in the previous project. The compounds will be designed to explore both structure activity relationships (SAR) and scope for structural modification to improve the compound profile (potency, DMPK, safety, etc.). Two series will be selected for Hit-to-Lead studies. The second 18-month phase of the project will involve the optimization of the series (prioritizing the series with the greatest potential) with the goal of identifying a series with a frontrunner compound meeting the MMV Early Lead criteria. |
| Project Detail | https://www.ghitfund.org/investment/portfoliodetail/detail/202/en |
T2021-256
| Project Title | Development of a Plasmodium vivax multistage vaccine effective both for protection and transmission blocking |
| Collaboration Partners | Hokkaido University, Jichi Medical University, Kanazawa University, Kyoto University, University of Toyama, University of Cambridge, Instituto Leônidas & Maria Deane (ILMD) and The Fundação de Medicina Tropical Doutor Heitor Vieira Dourado (FMT-HVD) |
| Disease | Malaria |
| Intervention | Vaccine |
| Stage | Antigen Identification |
| Awarded Amount | ¥69,817,000 (US$604,006) |
| Status | New project |
| Summary | [Project objective] The project aim is to develop not only a highly effective and durable multistage vaccine against pre-erythrocytic and sexual stages of P. vivax, but additionally, a bivalent vaccine effective both for P. vivax and P. falciparum.
[Project design] Two viral-vectored vaccines expressing both pre-erythrocytic-stage and sexual-stage antigens of P. vivax will be generated. Protective and transmission blocking (TB) efficacies of the heterologous prime-boost immunization regimen will be assessed by sporozoite challenge and Direct Membrane Feeding Assay (DMFA) in a robust and proven mouse model, and then the regime will be further optimized (e.g., dose, route, interval and outbred mice). Desired protection rate >90%. Surrogate markers responsible for protection will be identified. This will be key to allow efficient and robust measurements of efficacy. Humoral and cellular immune responses induced by the heterologous prime-boost immunization regimen will be assessed. Meanwhile, a bivalent vaccine harboring the genes encoding antigens of both P. vivax and P. falciparum will be generated. In a non-human primate model, in vitro sporozoite neutralizing assay and in vivo sporozoite challenge test of mice passively transferred with immune monkey IgGs will be performed to evaluate its protective efficacy. For evaluation of transmission blocking efficacy, immune monkey sera will be tested by DMFA using blood of vivax patients in Brazil and of falciparum patients in Burkina Faso. |
| Project Detail | https://www.ghitfund.org/investment/portfoliodetail/detail/200/en |
*All amounts are listed at the exchange rate of USD1 = JPY¥115.59, the approximate exchange rate on February 28, 2022.
Appendix.3 Investment Overview (As of March 31, 2022)
1. Investment to date
Total investments 27.6 billion yen (US$239 million*)
Total invested projects 114(active projects 61, completed projects 53)
*All amounts are listed at the exchange rate of USD1 = JPY¥115.59, the approximate exchange rate on February 28, 2022.
To know more about GHIT investments, please visit
Investment Overview: https://www.ghitfund.org/investment/overview/en
Portfolio: https://www.ghitfund.org/investment/portfolio/en
Advancing Portfolio: https://www.ghitfund.org/investment/advancingportfolio/en
Clinical Candidates: https://www.ghitfund.org/investment/clinicalcandidates/en
For more information, contact:
Katy Lenard at +1-301-280-5719 or [email protected]
Bumpei Tamamura at +81-36441-2032 or [email protected]
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/ghit-fund-announces-new-investments-a-total-of-740-million-yen-for-malaria-drugs-and-vaccine-301514408.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/ghit-fund-announces-new-investments-a-total-of-740-million-yen-for-malaria-drugs-and-vaccine-301514408.html
SOURCE Global Health Innovative Technology (GHIT) Fund